Allergic reactions to synthetic membranes in Haemodialysis
INTRODUCTION
Throughout the history of haemodialysis, several types of "allergic" adverse reactions have been described, appearing during the haemodialysis session. These reactions, which have not always been due to allergic mechanisms, are produced by the interaction of blood with the components of the extracorporeal blood circuit. They can be understood as a manifestation of the bio-incompatibility of materials used [1] [2] [3]. Its causes have been known, and for the most part have been corrected.
The first reactions were described in relation to the use of ethylene oxide to sterilise lines and dialysers [4] [5] [6]. Ethylene oxide has been replaced by other disinfection systems such as water vapour or gamma rays [6]. The first cellulosic membranes, Cuprophan type, were capable of activating the complement and causing hypoxia by leukocyte pulmonary sequestration [1] [7] [8] [9]. These membranes have evolved to asymmetric cellulose triacetate (ATA), with this complication disappearing [10]. The combined use of AN69 and ACEI membranes led to adverse reactions associated with an increase in bradykinins, which was solved by modifying the membrane to AN69-ST [11] [12] [13]. On the other hand, we cannot forget that adverse reactions related to intradialysis administered medication and dialysis fluid contaminants can appear, with a completely different epidemiology.
For years, reactions associated with the use of synthetic membranes, generally polysulfones, have been classified as "allergic", "hypersensitive" or "anaphylactic" [14] [15] [16] [17]. Several possible causes are being considered that are being corrected.
SYMPTOMS OF ALLERGIC REACTIONS TO SYNTHETIC HAEMODIALYSIS MEMBRANES.
Reactions associated with the use of synthetic membranes in haemodialysis can be early or late, acute or subacute, serious or less severe, which means that the classic clinical classification in type A/B is not useful. It can even make the diagnosis difficult.
The forms of presentation, from more to less frequent, are: dyspnoea, hypotension, bronchospasm, cough, vomiting and other digestive symptoms, chest pain, pruritus, urticaria, fever, headache and confusion. In the most severe cases, up to cardio-respiratory arrest and death have been described [14] [16] [17].
Eosinophilia and hypoxia are common [17]; thrombopenia may also be found [18]. In half of the cases, it appears in the first week of exposure to the causative dialyser [16]. However, this interval may be extended up to 36 months. In 2/3 of the cases, symptoms appear in the first 30 minutes of the haemodialysis session, but can appear at any time during the session [16]. Patients suffering from an acute, severe and early reaction, with dyspnoea and hypotension, are easily diagnosed. The problem lies in those with less specific and late symptoms, and who in many cases take time to be diagnosed or are not diagnosed, which means that this pathology is under-diagnosed.
Pruritus in haemodialysis patients is a frequent symptom and its causes are multiple [19]. Haemodialysis patients have been described in whom pruritus improved when the dialyser was changed. For example, in a series of nine haemodialysis patients treated with polysulfone or PMMA dialysers who presented persistent pruritus, not explained by other causes, after changing the dialyser to one with a cellulose triacetate membrane (ATA®), the pruritus improved in seven of them [20].
In haemodialysis patients, fever of allergic origin has been described, which can sometimes simulate an infection. The case of a patient with a feverish condition is described, which persisted for months until an infectious origin was ruled out and an allergic cause was suspected. When the dialyser was switched from polysulfone to cellulose triacetate (TAC) the patient's symptoms disappeared [21] .
Chest pain associated with intradialysis hypotension is another type of presentation of reactions associated with the use of synthetic membranes in haemodialysis. Delgado Córdova et al. [22] describe a patient who presented with episodes of hypotension and intradialytic chest pain. After changing to another dialyser, BG 2.1 U ® of polymethylmethacrylate (PMMA), the tolerance to the sessions improved and with the subsequent replacement to a CT membrane, she did not present hypotensive episodes again. In laboratory tests, she presented mild eosinophilia and thrombocytopenia that also disappeared with the change of the membrane. Another similar case is described by Martín-Navarro et al. [23]. This is an 86-year-old man, with significant comorbidity, who underwent dialysis with a Polyflux® dialyser and who presented episodes of hypotension and intradialysis symptoms for a month. Later, with exposure to various dialysers with synthetic membranes, he began with changing symptoms: Bronchospasm and severe arterial hypotension, generalised burning sensation and urticaria. When switching to cellulose triacetate (TAC) (Sureflux®), reactions associated with the use of synthetic membranes in haemodialysis did not present any longer.
The majority of publications about reactions associated with the use of synthetic membranes in haemodialysis deal with isolated cases, such as those we have presented [18] [22] [23] [24] [25] [26] [27] [28] [29] [30] [31] [32] [33] [34] [35] [36]. There are very few publications in which several cases are grouped together [14] [16] [17] .
Reactions associated with the use of synthetic membranes in haemodialysis should be suspected in patients on dialysis with synthetic dialysers and with acute or chronic intradialysis symptoms, not explained by other causes. Eosinophilia, increased serum tryptase, or hypocomplementemia help the differential diagnosis.
EPIDEMIOLOGY
Very few epidemiological studies have been published on reactions associated with the use of synthetic membranes in haemodialysis. In 1993, Simon et al [37] conducted a study with the objective of evaluating the incidence of anaphylactoid reactions in haemodialysis, with cellulosic and synthetic membranes and the role of ACE inhibitors in them: they studied 1,536 patients from 30 dialysis centres (122,694 sessions) during a 6-month period of observation. They found an annual incidence of 0.17/1000 sessions, with cellulosic membranes and 4.2/1000 sessions, with synthetic membranes. The proportion of "allergic reactions" was 10-20 times higher with synthetic membranes and 3-4 times higher in patients treated with ACE inhibitors. The prevalence of severe "allergic reactions" was 0.25% in the total population studied and 0.5% in patients treated with synthetic membranes.
In a study carried out in Spain between 2015 and 2017 and in which 1,561 patients were recorded, pertaining to 9 haemodialysis units, a prevalence of patients with reactions associated with the use of synthetic membranes in haemodialysis of 2.37 was found % [17].
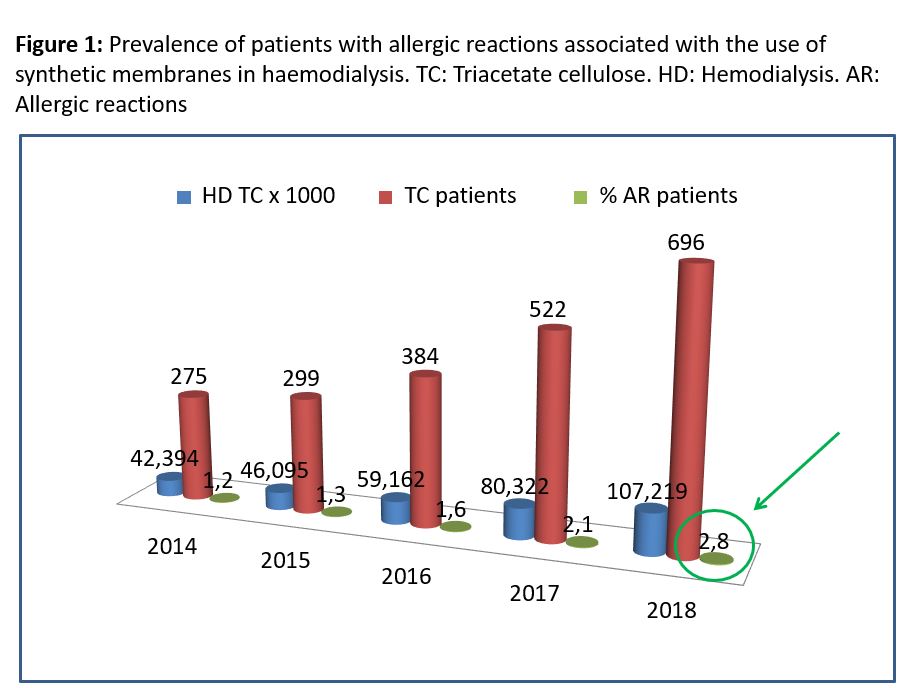
When studying the incidence/prevalence of reactions associated with the use of synthetic membranes in haemodialysis in a population on haemodialysis, it should be corrected for the number of patients, incidents or prevalent on haemodialysis and not based on haemodialysis sessions. This is so because the patient diagnosed with reactions associated with the use of synthetic membranes in haemodialysis begins to dialyse with time average concentration (TAC) and did not present allergic reactions again in the following sessions. In Spain, between 2014 and 2018, TAC dialysers were used exclusively to dialyse patients diagnosed with reactions associated with the use of synthetic membranes in haemodialysis. On a very few occasions, PMMA® or Evodial® membranes were used. With the data on CT consumption in Spain during these years and the prevalence data on haemodialysis from REER, we have calculated the prevalence of patients with reactions associated with the use of synthetic membranes in haemodialysis. In (Figura 1) we observe how until 2018, prevalence increased to 2.8%. This proportion is similar to that found by Esteras et al. [17].
CAUSES OF ALLERGIC REACTIONS TO SYNTHETIC MEMBRANES IN HAEMODIALYSIS
Allergic reactions to synthetic haemodialysis membranes have been described with the use of different membranes: polysulfone, polyethersulfone, polynephron, polyarylether sulfone, and polyacrylonitrile [16] [17] . In some of the published cases, the patient had been dialysed with several of these membranes, presenting reactions associated with the use of synthetic membranes in haemodialysis with all of them, with different symptoms. When the patient went to dialysis with TAC, he/she had no further reactions [23].
In the last 20 years, changes have been made to polysulfone membranes to improve their performance, but their biocompatibility has worsened. It has been described that reactions associated with the use of synthetic membranes in haemodialysis would present more frequently with polysulfones, but it should be borne in mind that these are currently the most used membranes. All membranes involved in reactions associated with the use of synthetic membranes in haemodialysis have in common that they contain polyvinylpyrrolidone (PVP). This substance is incorporated into the hydrophobic membrane to create hydrophilic spaces and improve hydraulic permeability. The amount of PVP and its location on the membrane varies from one synthetic membrane to another. Other substances, such as bisphenol A (BPA), could be involved, but not all affected dialysers contain it. Polysulfone adsorbs proteins that participate in blood-membrane interaction, such as ficolin 2, fibrinogen fragments, and proteins from red blood cells, which can contribute to complement activation, leukocyte adhesion, and blood clotting. Contrary to other membranes, TAC lacks PVP and bisphenol A. Furthermore, it is capable of adsorbing a large amount of albumin and apolipoproteins, increasing its biocompatibility and reducing platelet aggregation. New synthetic membranes are being developed without PVP or with structural modifications that could improve their biocompatibility [38] [39].
MECHANISMS OF ALLERGIC REACTIONS TO SYNTHETIC MEMBRANES IN HAEMODIALYSIS
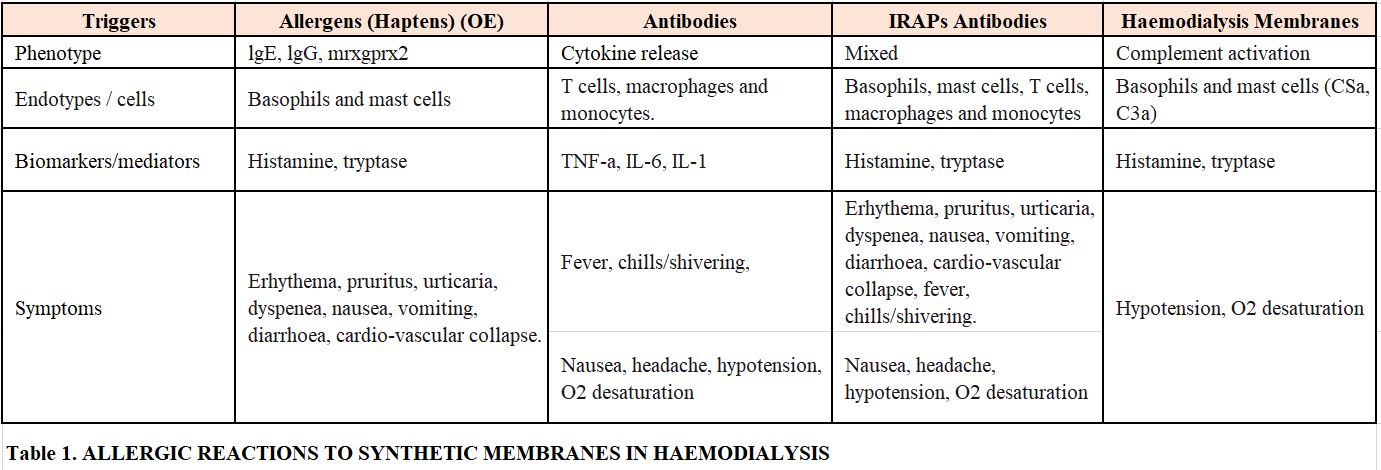
It is understood as a hypersensitivity reaction, which occurs when the immune system reacts inappropriately and causes tissue damage. Anaphylaxis is a systemic reaction caused by the release of mediators after degranulation of mast cells and basophils. Anaphylaxis may be due to an immunological trigger, dependent on IgE, IgG or activation of the complement or a non-immune trigger - MRGPRX2 [40][41].
The presence of activated basophils and T lymphocytes in peripheral blood of patients with hypersensitivity reactions to polysulfone has been demonstrated during the acute reaction [42]. It has also been shown that basal tryptase concentrations are elevated in patients allergic to this membrane [42]. Rodríguez-Sanz A et al. conclude in their study that the degranulation of basophils and elevated levels of tryptase, which occur during the acute reaction in patients allergic to synthetic membranes, demonstrate the activation of mast cells and basophils [42]. On occasions, it would be a type I specific anaphylactic reaction, mediated by IgE-basophil or by complement activation; on other occasions, it would be due to the direct degranulation of basophils (Tabla 1) . In some patients, complement activation is detected.
Several mediators could be activated, which would explain the variable symptoms presented by these patients. An exposed or eluted component of the membrane would be responsible as an adjuvant of cell activation in patients with reactions associated with the use of synthetic membranes in haemodialysis [42].
TREATMENT
Acute and severe symptoms should be treated by stopping the blood pump and disconnecting the lines without returning the blood to the patient. At the same time, symptomatic treatment should be started: oxygen therapy, intravenous steroids, antihistamines, and bronchodilators as appropriate. If the patient stabilises, dialysis could be restarted by switching to a CT dialyser. In less severe cases, diagnosis and treatment is "ex juvantibus" by changing to a CT dialyser. Currently, there are high-performance, HDF-OL capable CT dialysers [10] .
CONCLUSIONS
Currently, there is a higher incidence of hypersensitivity reactions in haemodialysis associated with synthetic membranes.
There should be suspicion when acute or chronic symptoms appear during the haemodialysis session, which cannot be explained by other causes.
The diagnosis and treatment is "ex juvantibus," checking results when changing to a cellulose triacetate membrane.